Understanding Anxiety and Depression Medication Side Effects Comparison: How Your Prescription Might Be Stealing Your Sleep
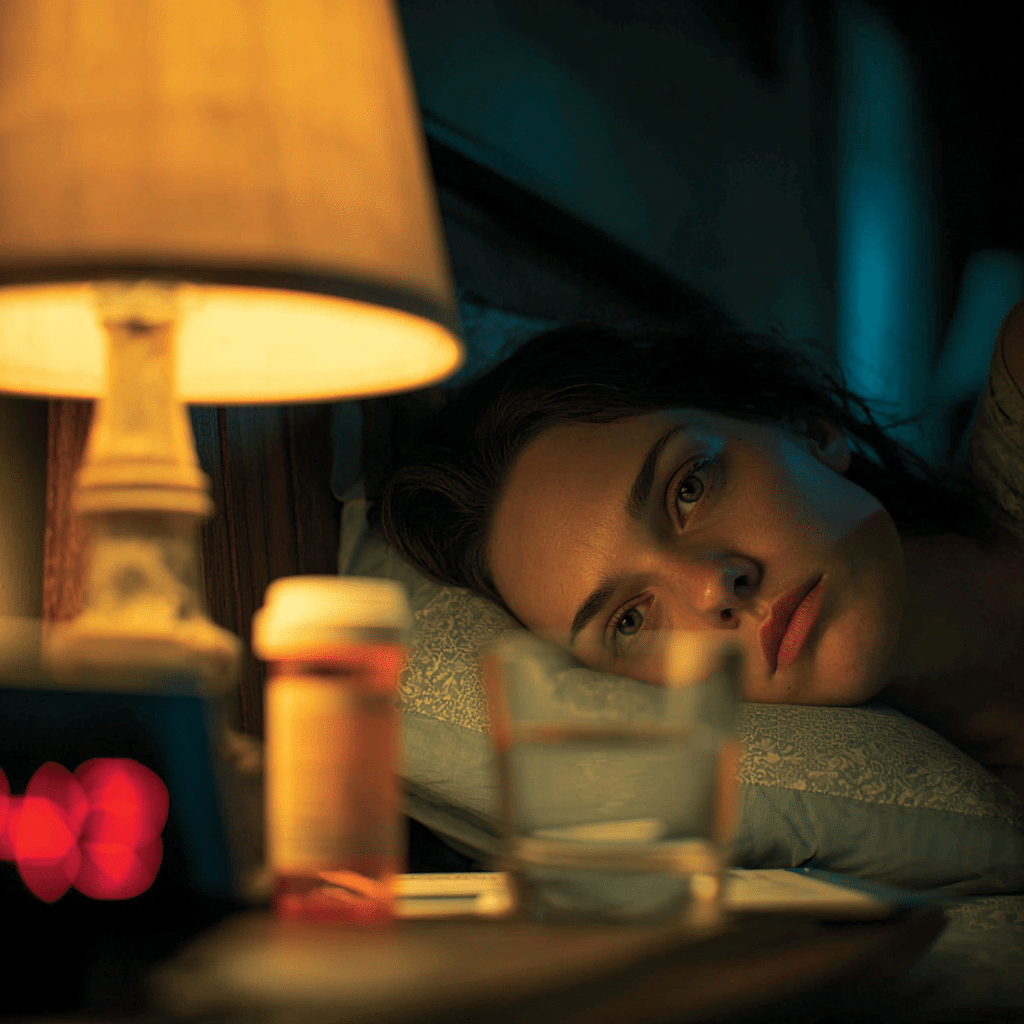
Story-at-a-Glance
- Sleep disruption is among the most common and troublesome long-term side effects of anxiety and depression medications, affecting up to 60% of patients on SSRIs/SNRIs despite successful mood treatment
- Different medication classes have vastly different sleep impacts: SSRIs and SNRIs commonly cause insomnia and suppress REM sleep, while benzodiazepines reduce sleep quality despite helping you fall asleep faster
- The relationship between dose and sleep side effects isn’t always linear—some medications like mirtazapine are more sedating at lower doses, creating a complex treatment puzzle
- A comprehensive 2023 analysis of 21 antidepressants found reboxetine and vilazodone had the highest insomnia risk, while fluvoxamine, trazodone, and mirtazapine topped the list for causing excessive sleepiness
- Recent medication approvals signal hope: January 2025 saw Spravato approved as monotherapy for treatment-resistant depression, while novel treatments targeting sleep and mood simultaneously entered late-stage trials
- The “best” medication isn’t the one with fewest side effects—it’s the one that treats your underlying condition while minimizing sleep disruption specific to your situation
When Sarah finally got her anxiety under control with sertraline, she thought her sleepless nights would end. Instead, they got worse. Three months into treatment, she lay awake most nights despite feeling calmer during the day. Her psychiatrist wasn’t surprised—approximately 60% of patients on standard SSRIs and SNRIs experience residual insomnia symptoms, even when their mood symptoms improve significantly.
This presents a frustrating paradox. You’re taking medication to feel better, yet the very pills that lift your mood might be sabotaging your sleep. Understanding the anxiety and depression medication side effects comparison becomes crucial when sleep problems emerge during treatment.
The Sleep-Medication Paradox
Here’s what makes this situation particularly challenging: depression and anxiety themselves disrupt sleep. But so do many medications used to treat them. Research from the Institute of Psychiatry and Neurology in Warsaw reveals that sleep disturbances are not only symptoms of depression but also the most frequent residual symptoms after treatment. This means they often persist even when other symptoms improve.
Dr. Adam Wichniak, a leading sleep medicine researcher at the Institute of Psychiatry and Neurology, has spent years documenting how antidepressants affect sleep architecture. His work demonstrates that these effects depend not just on which drug you take, but also on the dose and timing of administration. Duration of treatment matters too. It’s far more nuanced than “this medication causes insomnia” or “that one makes you sleepy.”
The reality is more complex. Some medications suppress REM sleep (the stage associated with dreaming and memory consolidation). Others reduce slow-wave sleep (the deepest, most restorative stage). Still others improve sleep continuity but cause next-day grogginess that affects your quality of life.
Breaking Down Medication Classes and Their Sleep Effects
SSRIs: The Most Prescribed, Most Complicated
Selective Serotonin Reuptake Inhibitors remain first-line treatment for both anxiety and depression. Common names include fluoxetine (Prozac), sertraline (Zoloft), escitalopram (Lexapro), paroxetine (Paxil), and citalopram (Celexa).
How they affect sleep:
According to a comprehensive 2023 network meta-analysis published in the journal SLEEP, SSRIs have a paradoxical relationship with sleep. This analysis examined data from over 64,000 patients across 216 randomized controlled trials. During initial treatment (the first 2-4 weeks), they frequently cause or worsen insomnia. Clinical observations show that SSRIs increase sleep latency (time to fall asleep) and suppress REM sleep. They may also fragment sleep throughout the night.
The catch? If the medication successfully treats your depression, your overall sleep often improves after 3-4 weeks—but not always. Studies indicate that 30-40% of patients on maintenance treatment with SSRIs continue experiencing insomnia.
Among SSRIs, there are notable differences. Surveillance data suggests that fluoxetine and sertraline are most often associated with anxiety, agitation, and insomnia. Citalopram appears better tolerated overall. However, individual responses vary dramatically.
Timeline matters: Most sleep-disrupting effects are strongest in the first few weeks. Some people adapt; others don’t. The question becomes: how long do you wait to see if sleep improves?
SNRIs: Double the Neurotransmitters, Double the Activation?
Serotonin-Norepinephrine Reuptake Inhibitors like venlafaxine (Effexor), duloxetine (Cymbalta), and desvenlafaxine (Pristiq) affect both serotonin and norepinephrine.
The 2023 SLEEP analysis found that desvenlafaxine ranked among the top medications for insomnia risk. This makes physiological sense: norepinephrine is a “wake-promoting” neurotransmitter. By increasing its availability, SNRIs can increase alertness—which is great during the day but problematic at night.
According to research from The Better Sleep Clinic, venlafaxine and sertraline are the most “activating” medications they see in clinical practice. Patients frequently report increased wakefulness and difficulty maintaining sleep.
Additionally, some SNRIs carry a risk of blood pressure elevation, which can further interfere with sleep quality. Yet for certain conditions—particularly chronic pain syndromes that coexist with depression—SNRIs may be the most effective choice. This remains true despite sleep side effects.
Benzodiazepines: Fast Relief, Hidden Costs
Benzodiazepines (alprazolam/Xanax, lorazepam/Ativan, clonazepam/Klonopin, diazepam/Valium) work quickly for anxiety and help you fall asleep faster. They’re prescribed for short-term anxiety relief, panic attacks, and sometimes as a bridge when starting other medications.
Here’s the problem: while benzodiazepines make you fall asleep faster and sleep longer, they fundamentally alter sleep architecture. These changes reduce sleep quality in significant ways.
Research published in Frontiers in Psychiatry reveals that benzodiazepines increase light sleep (Stage 2). However, they decrease slow-wave sleep—the deep, restorative sleep your brain needs. Think of it as sleeping eight hours but getting the restorative benefit of only five or six.
The bigger concern? Physical dependence can develop within 2-4 weeks of daily use. American Academy of Family Physicians research emphasizes that withdrawal symptoms are possible after just one month. Discontinuation can trigger severe rebound insomnia—often worse than the original sleep problem.
A comprehensive review from the National Center for Biotechnology Information documented that regular benzodiazepine use has been linked to cognitive decline and increased fall risk, especially in elderly patients. Paradoxically, in some cases, it also increased anxiety and aggression.
Yet benzodiazepines remain widely prescribed. Why? Because for acute situations—panic attacks, severe anticipatory anxiety, temporary crisis situations—they work, and they work fast. The key phrase is “short-term.”
Medications That Actually Promote Sleep
Not all anxiety and depression medications disrupt sleep. Some actively promote it.
Low-dose doxepin is actually FDA-approved for insomnia at doses of 3-6 mg—far below the antidepressant dose of 75-300 mg. At these low doses, it blocks histamine receptors (similar to Benadryl but more selective). This helps maintain sleep without significant next-day sedation.
Trazodone has become one of the most commonly prescribed sleep aids despite not being FDA-approved for insomnia. Research shows it reduces the number of nighttime awakenings and helps people fall asleep within 30 minutes. However, some people build tolerance to its sedative effects over time.
Mirtazapine presents an interesting dose-response curve. It’s more sedating at lower doses (7.5-15 mg) because at these doses, histamine blockade dominates. At higher doses (30-45 mg), norepinephrine effects increase, reducing sedation while increasing antidepressant efficacy. Sleep Foundation research confirms this paradoxical pattern—making dose titration tricky.
What the Latest Research Reveals About Individual Medications
The 2023 Oxford Academic network meta-analysis—the most comprehensive study to date—ranked 21 antidepressants. The ranking focused on their likelihood of causing insomnia or excessive sleepiness (somnolence).
Highest insomnia risk:
- Reboxetine (odds ratio 3.47 times higher than placebo)
- Vilazodone
- Desvenlafaxine
Highest somnolence risk:
- Fluvoxamine (odds ratio 6.32 times higher than placebo)
- Trazodone
- Mirtazapine
Notably, medications with minimal sleep side effects included vortioxetine—one of the newer antidepressants that showed no significantly higher risk for either somnolence or insomnia compared to placebo in this analysis.
But here’s where it gets interesting: the relationship between dose and sleep side effects varied dramatically between medications. Some showed linear relationships (more medication = more side effect), while others showed inverted U-shapes or more complex patterns. This suggests that for some medications, adjusting the dose up or down might reduce sleep problems.
Save This Article for Later – Get the PDF Now
Current Developments: Hope on the Horizon
The pharmaceutical landscape is evolving. In January 2025, the FDA expanded approval of Spravato (esketamine) to be used as monotherapy. This means it can now be prescribed alone without requiring an oral antidepressant. This matters for sleep because traditional antidepressants often contribute to insomnia in treatment-resistant depression.
Even more promising: Johnson & Johnson’s Phase 3 trials of seltorexant—an orexin-2 receptor antagonist—demonstrated simultaneous improvement in both depressive symptoms and sleep disturbance. These results came from patients who hadn’t responded adequately to SSRIs/SNRIs. This represents a potential paradigm shift. Instead of treating mood and sleep as separate problems requiring separate solutions, one medication might address both.
In November 2025, CAPLYTA (lumateperone) gained FDA approval as an adjunctive therapy for major depressive disorder. It offers a significant advantage: not causing the weight gain, metabolic changes, or sexual side effects that often lead patients to discontinue treatment.
Making Sense of Your Situation
Given all these variables, how do you and your healthcare provider make decisions?
The top sleep specialists would emphasize several key principles:
1. Treating the underlying condition is priority one. An untreated mood or anxiety disorder disrupts sleep more severely than most medication side effects. The goal is optimal treatment of your primary condition with minimal sleep disruption—not zero side effects.
2. Individual response trumps population statistics. Some people take fluoxetine (known for causing insomnia) and sleep better than ever. Others take mirtazapine (known for sedation) and still can’t sleep. You might be an outlier, and that’s okay.
3. Timing matters enormously. Taking an activating medication at bedtime is asking for trouble. Dr. Wichniak’s research emphasizes that the time of administration can be as important as which medication you choose. Morning dosing of SSRIs/SNRIs often reduces nighttime insomnia.
4. Give it time—but not too much time. Most acute side effects improve after 2-4 weeks. But if you’re still experiencing significant sleep disruption after 6-8 weeks, it’s reasonable to discuss alternatives.
5. Combination approaches work. Many psychiatrists use a small dose of a sedating antidepressant (like trazodone or mirtazapine) alongside a primary antidepressant. This addresses both mood and sleep. As noted in GreatSleep.blog’s guide to antidepressants for insomnia, this strategy can be particularly effective for long-term insomnia management.
When Medication Isn’t Enough
Sometimes, even with optimal medication management, sleep problems persist. This is where the anxiety and depression medication side effects comparison becomes less about choosing different pills. It becomes more about comprehensive treatment.
Research published in the Cochrane Database found limited evidence supporting long-term antidepressant use specifically for insomnia. This suggests that medication alone may not be sufficient. The review emphasized that despite widespread prescribing of antidepressants for sleep problems, the evidence remains surprisingly limited. High-quality research supporting this practice is lacking.
Cognitive Behavioral Therapy for Insomnia (CBT-I) has robust evidence as a first-line treatment. When combined with appropriate medication management, outcomes improve significantly. Sleep hygiene, light therapy, and addressing lifestyle factors all play roles.
But let’s be honest: telling someone with severe depression to “practice good sleep hygiene” feels dismissive. The point isn’t to abandon medication—it’s to recognize that medication works best as part of a comprehensive approach.
What About Long-Term Use?
This question deserves its own consideration. Most clinical trials last 8-12 weeks. Real-world use often extends for years.
Long-term effects of SSRIs include sexual dysfunction, weight gain, and sleep disturbance. Sleep disturbance persists in a significant subset of patients. The question becomes: is continued medication use improving your quality of life overall? Or has it become part of the problem?
For benzodiazepines, European guidelines recommend use no longer than 4 weeks due to tolerance, dependence, and declining efficacy. Yet many patients remain on them for months or years, often because withdrawal is so challenging.
There’s no simple answer. Chronic anxiety or depression often requires chronic treatment. The key is ongoing assessment: Is this still working? Are the benefits outweighing the costs?
A Word About Withdrawal and Discontinuation
If you and your healthcare provider decide to change medications due to sleep side effects, the way you stop matters. It matters as much as what you take next.
Antidepressant discontinuation syndrome can cause severe insomnia, among other symptoms. Gradual tapering over several weeks (or months for long-term use) reduces these risks significantly.
Benzodiazepine withdrawal can be even more severe—and potentially dangerous if done abruptly after prolonged use. Medical supervision is essential for benzodiazepine discontinuation.
Never stop psychiatric medications suddenly or without medical guidance. This isn’t about medical liability—it’s about your safety and comfort.
Finding Your Individual Solution
The anxiety and depression medication side effects comparison ultimately isn’t about which medication is “best” in general. It’s about which approach works best for you, right now. Your specific constellation of symptoms, medical history, and treatment goals all matter.
Consider these questions for discussion with your healthcare provider:
- Is sleep disruption from the medication better or worse than sleep disruption from untreated anxiety/depression?
- Have you tried adjusting timing (morning vs. evening dosing)?
- Would a different medication in the same class work better?
- Would adding a sleep-promoting medication be more effective than switching?
- Are non-medication interventions being utilized effectively?
The fact that you’re researching this topic suggests you’re engaged in your treatment—that’s valuable. Bring your observations, concerns, and questions to your healthcare provider. The best treatment decisions happen through informed collaboration.
Remember: medications are tools, not solutions in themselves. They create conditions that make recovery possible—but recovery itself requires time, often therapy, and usually lifestyle changes as well.
Your sleep matters. Your mood matters. Finding the balance between treating one without sacrificing the other is possible—it just might take some trial, adjustment, and patience to get there.
FAQ
Q: What are SSRIs and SNRIs?
A: SSRIs (Selective Serotonin Reuptake Inhibitors) are antidepressants that work by increasing serotonin levels in the brain. Common examples include Prozac, Zoloft, and Lexapro. SNRIs (Serotonin-Norepinephrine Reuptake Inhibitors) increase both serotonin and norepinephrine, like Effexor and Cymbalta. Both classes are first-line treatments for depression and anxiety disorders.
Q: What is REM sleep and why does it matter?
A: REM (Rapid Eye Movement) sleep is a sleep stage characterized by rapid eye movements, vivid dreams, and heightened brain activity. It’s critical for memory consolidation, learning, and emotional processing. Many antidepressants suppress REM sleep, which may affect these cognitive functions, though the clinical significance remains debated.
Q: What does “sleep architecture” mean?
A: Sleep architecture refers to the structure and pattern of sleep stages throughout the night. Normal sleep cycles through NREM (non-rapid eye movement) stages 1-3 and REM sleep multiple times per night. Medications can alter this architecture—for example, reducing deep slow-wave sleep or suppressing REM sleep—which affects sleep quality even if total sleep time seems adequate.
Q: What is treatment-resistant depression?
A: Treatment-resistant depression (TRD) is major depressive disorder that hasn’t responded adequately to at least two different antidepressant treatments at adequate doses and duration. It affects approximately one-third of people with depression and often requires alternative or combination treatment approaches.
Q: What does “off-label” prescription mean?
A: Off-label means a doctor prescribes a medication for a use not specifically approved by the FDA, even though the medication itself is FDA-approved for other conditions. For example, trazodone is FDA-approved as an antidepressant but is commonly prescribed off-label for insomnia. This practice is legal and often evidence-based.
Q: What is slow-wave sleep?
A: Slow-wave sleep (SWS), also called deep sleep or Stage 3 NREM sleep, is the most physically restorative sleep stage. During SWS, your body repairs tissues, strengthens the immune system, and consolidates certain types of memories. Some antidepressants reduce the amount of slow-wave sleep, potentially affecting these restorative processes.
Q: What are benzodiazepines and are they addictive?
A: Benzodiazepines are anti-anxiety medications that enhance the effects of GABA, a calming neurotransmitter. They include medications like Xanax, Ativan, and Valium. While effective for short-term anxiety relief, they carry significant risk of physical dependence with regular use—your body adapts to their presence, and stopping them abruptly can cause withdrawal symptoms. This is different from addiction (compulsive use despite harm) but remains a serious concern.
Q: What is sleep latency?
A: Sleep latency is the amount of time it takes you to fall asleep after turning off the lights and intending to sleep. Normal sleep latency is 10-20 minutes. Many activating antidepressants increase sleep latency, meaning it takes longer to fall asleep.
Q: How does this article’s information about anxiety and depression medication side effects comparison help me make treatment decisions?
A: This comparison helps you understand that medication side effects vary significantly between drug classes and even between individuals. The article emphasizes that there’s no universally “best” medication—the optimal choice depends on your specific symptoms, medical history, and how you personally respond. It provides a framework for informed discussions with your healthcare provider about balancing mood improvement with sleep quality, and highlights that timing, dosage, and combination approaches all matter in optimizing treatment outcomes.

